 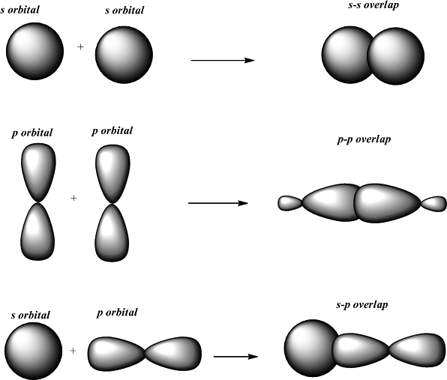
The structure has one six membered ring fused with the one five membered ring and totally has alternate carbon and nitrogen bonds wherein two oxygen atoms are present on the two carbon atoms of the six membered ring as a carbonyl group. Which two sets of reactants best represents (c) one sp 2-sp2 sigma bond, one p-p pi bond the amphoteric. 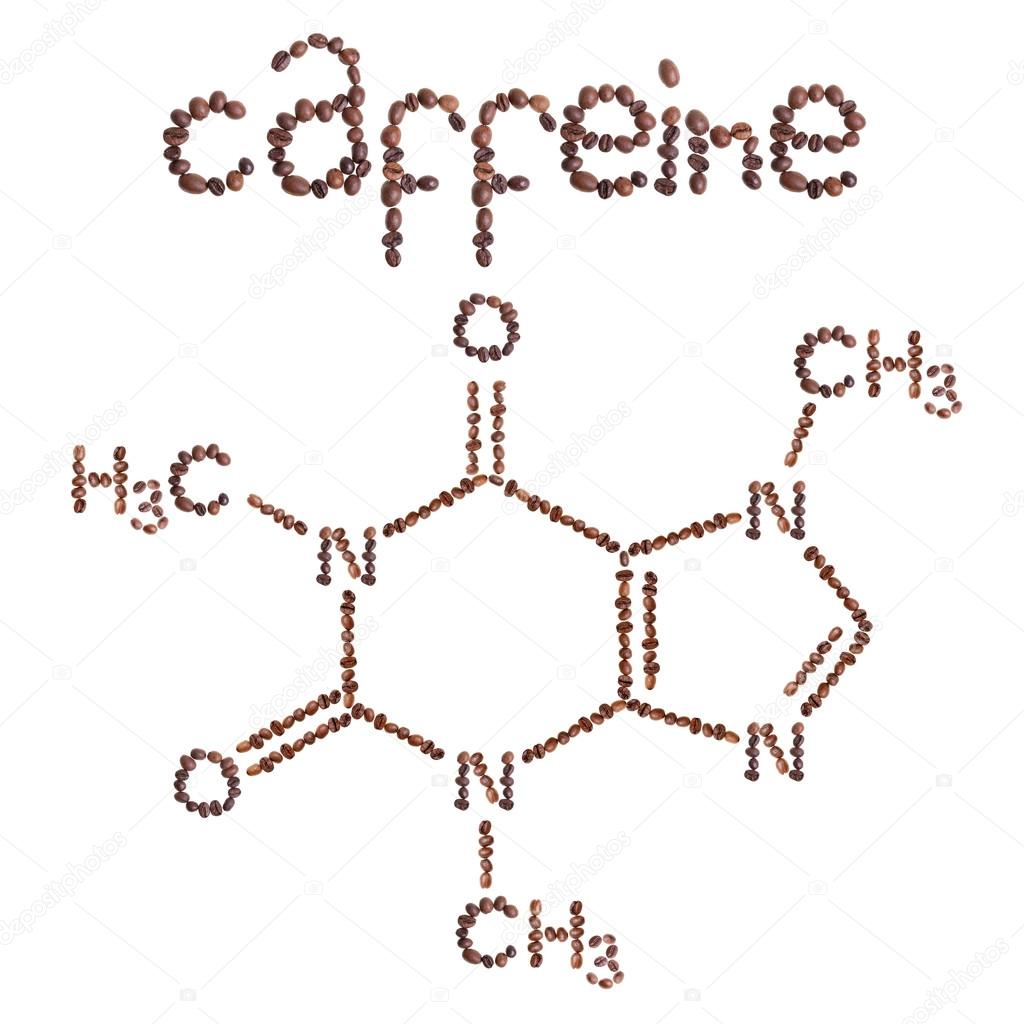
Hint: The concept of organic chemistry gives the answer where writing down the structure of caffeine which has molecular formula $$ where it has two cyclic structures attached to each other that consist of the hetero atoms. How many pi bonds are present in caffeine How many sigma bonds Insert the lone pairs in the molecule. Caffeine has a molecular weight of 194 u. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
